Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Home Learning Center Citric Acid Anhydrous
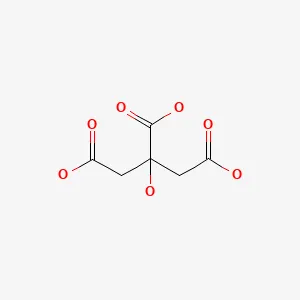
Citric Acid Anhydrous Structure
| IUPAC Name | : 2-hydroxypropane-1,2,3-tricarboxylic acid |
| CAS Number | : 77-92-9 |
| HS Code | : 2918.14 |
| Molecular Formula | : C6H8O7 |
| Structural Formula | : HOOCCH2C(OH)(COOH)CH2COOH |
| Synonyms | : Citric acid, Anhydrous citric acid, 2-Hydroxytricarballylic acid, E330 |
| Molecular Weight | : 192.12 g/mol |
Citric acid anhydrous is a weak organic tricarboxylic acid naturally present in citrus fruits. It is one of the most widely used food additives globally, approved under E330, and valued for its sharp, clean acidic taste, excellent solubility, and broad functional versatility across food, beverage, pharmaceutical, and industrial applications.
As an anhydrous form, it contains no water of crystallization, making it more stable, easier to handle in dry blending, and more concentrated compared to citric acid monohydrate. Its high purity, consistent quality, and GRAS (Generally Recognized as Safe) status make it a preferred acidulant in food manufacturing worldwide.
Commercially, citric acid is produced through fermentation of sugars by Aspergillus niger mold, making it a bio-based and renewable ingredient. It plays critical roles as an acidulant, preservative, chelating agent, pH regulator, and flavor enhancer across a vast range of applications.
| Physical Description | : White crystalline powder or granules. |
| Color / Form | : White to off-white. |
| Odor | : Odorless. |
| Taste | : Strong, clean sour/acidic taste. |
| Boiling Point | : Decomposes above 175 °C |
| Melting Point | : 153–159 °C |
| Flash Point | : Not applicable (solid) |
| Solubility | : Freely soluble in water; soluble in ethanol. |
| Density | : Approx. 1.665 g/cm³ |
| pH (1% solution) | : Approx. 2.2 |
| Stability / Shelf Life | : Stable under normal dry storage conditions. Hygroscopic above 75% RH. |
| Viscosity | : N/A (solid form) |
| Polymerization | : No hazardous polymerization. |
Citric acid anhydrous is one of the most versatile food additives in use today. In beverages, it is the primary acidulant in carbonated soft drinks, fruit juices, energy drinks, and powdered drink mixes, providing tartness and enhancing fruit flavor profiles. It also acts as a preservative by lowering pH and inhibiting microbial growth, extending product shelf life.
In confectionery, citric acid gives sour candies, gummies, and hard candies their characteristic sharp taste. It is used in jam and jelly production to adjust pH for optimal pectin gelation, and in dairy products such as processed cheese and cream cheese as an emulsifying salt and pH regulator. In bakery applications, it acts as a leavening acid when combined with sodium bicarbonate.
Beyond food and beverages, citric acid is a key chelating agent in pharmaceutical formulations, cleaning products, and cosmetics. It sequesters metal ions that would otherwise catalyze oxidative degradation, improving stability of sensitive formulations. In industrial applications, it is used in descaling agents, metal cleaning, and textile processing.
Commercial production of citric acid is based almost exclusively on submerged fermentation of carbohydrate substrates (typically molasses, glucose, or sucrose) using the filamentous fungus Aspergillus niger. The fermentation is conducted in large stirred-tank or airlift bioreactors under controlled temperature, aeration, and pH conditions optimized for maximum citric acid yield.
After fermentation, the citric acid is recovered from the broth by precipitation with lime (calcium hydroxide) to form calcium citrate, followed by acidulation with sulfuric acid to release free citric acid. The resulting crude citric acid solution is purified by activated carbon treatment, ion exchange, and evaporative crystallization to produce high-purity food-grade or pharmaceutical-grade product. The anhydrous form is produced by crystallization above 36.6 °C, while the monohydrate form crystallizes below this temperature.
Citric acid anhydrous is of low acute toxicity and is generally recognized as safe (GRAS) for food use. It may cause irritation to skin, eyes, and respiratory tract on direct contact or inhalation of dust.
Combustible solid. Dust may form ignitable mixtures with air at elevated concentrations. Not classified as a significant fire risk under normal storage conditions.
Dust or concentrated solutions may cause mild to moderate irritation of skin, eyes, and mucous membranes. Prolonged skin contact with concentrated solutions may cause mild irritation or dryness.
Combustible solid; fine dust may form explosive mixtures in air.
Wash affected area thoroughly with soap and water. Remove contaminated clothing. Seek medical advice if irritation persists.
Flush immediately with clean water for at least 15 minutes, lifting eyelids occasionally. Seek medical attention if irritation persists.
Rinse mouth with water. Give water to drink. Seek medical advice if large quantities are ingested or symptoms appear.
Use CO², dry chemical powder, or foam extinguisher. Water spray may be used to cool containers. Avoid generating dust during firefighting.
Small spill: Sweep or vacuum up carefully to avoid generating dust. Collect in labeled, sealable containers. Do not wash down drains — citric acid has high BOD in waterways.
Large spill: Contain and collect mechanically. Prevent entry into drains and waterways. Dispose in accordance with local regulations.
Store in original, tightly sealed packaging in a cool, dry, well-ventilated area. Citric acid anhydrous is hygroscopic — exposure to humidity above 75% RH causes caking and conversion to monohydrate form. Keep away from strong oxidizing agents, strong bases, and metal powders. Store away from food contact surfaces if not food-grade handling.
Recommended storage temperature: 15–25 °C. Relative humidity: below 60% RH. Shelf life: 24–36 months in original sealed packaging. Suitable containers: food-grade HDPE, multi-wall paper bags with PE liner, or fiber drums. Protect from moisture, direct sunlight, and heat sources. FIFO rotation recommended.
For more detailed information including pricing, customization, and shipping:
Discover more about this product and understand how it may fit into your operations, formulations, or production requirements.
Browse Product