Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Home Learning Center Lactic Acid
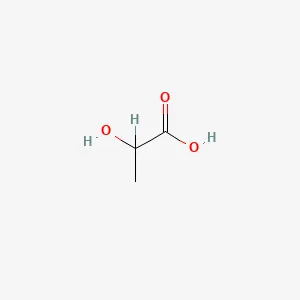
Lactic Acid Structure
| IUPAC Name | : 2-hydroxypropanoic acid |
| CAS Number | : 50-21-5 |
| HS Code | : 2918.11 |
| Molecular Formula | : C3H6O3 |
| Structural Formula | : CH3CH(OH)COOH |
| Synonyms | : DL-Lactic acid, 2-Hydroxypropionic acid, Milk acid, E270 |
| Molecular Weight | : 90.08 g/mol |
Lactic acid is a naturally occurring organic acid found in milk, fermented foods, and produced metabolically in the human body during exercise. As a food additive (E270), it is widely used as an acidulant, preservative, and flavoring agent in a broad range of food and beverage products.
Commercial lactic acid is available in both L(+) and D(-) enantiomeric forms, as well as the racemic DL mixture. The L(+) form is preferred in food applications as it is the naturally occurring biological form. It is supplied as an aqueous solution, typically at 80% or 88% concentration.
Beyond food, lactic acid is an important building block for polylactic acid (PLA), a biodegradable bioplastic, and is widely used in personal care, pharmaceutical, and industrial cleaning applications. Its bio-based, biodegradable nature makes it a preferred choice in sustainable chemistry initiatives.
| Physical Description | : Clear, colorless to slightly yellow viscous liquid. |
| Color / Form | : Colorless to pale yellow liquid. |
| Odor | : Mild, characteristic sour odor. |
| Taste | : Sour, acidic taste. |
| Boiling Point | : Approx. 122 °C at 15 mmHg (decomposes at higher temperatures) |
| Melting Point | : 16.8 °C (pure); typically liquid at room temperature as aqueous solution |
| Flash Point | : >110 °C |
| Solubility | : Miscible with water, ethanol, and acetone. |
| Density | : Approx. 1.20 g/cm³ (80% solution) |
| pH (1% solution) | : Approx. 2.4–2.5 |
| Stability / Shelf Life | : Stable under normal storage. May self-condense to lactide or polylactic acid at elevated temperatures. |
| Viscosity | : Approx. 40–70 mPa·s at 25 °C (80% solution) |
| Polymerization | : May self-condense at high temperatures; no hazardous polymerization under normal conditions. |
In food and beverage applications, lactic acid is used as an acidulant, preservative, and flavor enhancer. It is widely used in dairy products (cheese, yogurt, cultured butter), fermented meats, pickled vegetables, salad dressings, sauces, and beverages. Its mild, smooth acidity is preferred over sharper acids in many dairy and fermented product applications.
As a preservative, lactic acid inhibits the growth of pathogenic and spoilage microorganisms by lowering pH and dissociating into the undissociated acid form, which penetrates microbial cell membranes. It is used in combination with other hurdle technologies in ready-to-eat meats, seafood, and minimally processed foods. Sodium lactate and potassium lactate (salts of lactic acid) are also widely used as antimicrobial agents and humectants in processed meat products.
In personal care, lactic acid is used as a skin-conditioning agent, pH adjuster, and alpha-hydroxy acid (AHA) in exfoliant and anti-aging skin care products. In pharmaceutical applications, it is used in topical formulations, parenteral solutions, and as a component of lactated Ringer's solution. The industrial application of lactic acid includes use as a descaling agent, solvent, and monomer for PLA bioplastic production.
Commercial lactic acid is produced primarily by fermentation of carbohydrate substrates (glucose, sucrose, lactose, or starch hydrolysates) using homofermentative lactic acid bacteria (LAB) such as Lactobacillus species. The fermentation is conducted under controlled temperature (40–45 °C), pH (5.5–6.5 maintained with calcium carbonate or sodium hydroxide), and anaerobic conditions to maximize lactic acid yield and minimize by-product formation.
After fermentation, crude lactic acid is recovered by acidification of calcium or sodium lactate, followed by filtration, activated carbon treatment for decolorization, evaporation, and purification by ion exchange or distillation. Food- and pharmaceutical-grade lactic acid requires high purity, low residual sugar, and absence of heavy metals. The L(+) form is produced using enantioselective Lactobacillus strains, while chemical synthesis routes (from acetaldehyde) produce the racemic DL mixture.
Lactic acid is of low acute toxicity at food-use concentrations. Concentrated solutions (80–88%) may cause irritation to skin, eyes, and mucous membranes. Not classified as carcinogenic or mutagenic.
Combustible liquid with a relatively high flash point (>110 °C). Low fire risk under normal storage and handling conditions.
Concentrated lactic acid solutions may cause skin and eye irritation on direct contact. Vapor at elevated temperatures may irritate the respiratory tract.
Combustible liquid at elevated temperatures.
Remove contaminated clothing. Wash affected area with soap and water for at least 15 minutes. Seek medical advice if irritation persists.
Flush immediately with clean water for at least 15 minutes. Seek medical attention.
Rinse mouth with water. Give water to drink. Seek medical advice if large quantities of concentrated product are ingested.
Use CO², foam, or dry chemical. Water spray may be used to cool containers. Avoid direct water jet on burning liquid.
Small spill: Absorb with inert absorbent material (sand, earth, or commercial absorbent). Collect in labeled, sealable containers. Rinse residual area with water. Prevent entry into drains.
Large spill: Contain with bunds. Do not allow entry into waterways (high BOD). Pump into recovery containers. Clean residual with water. Notify environmental authorities for significant spills.
Store in original, tightly closed containers made of HDPE, stainless steel 304/316, or glass-lined vessels. Keep away from strong oxidizing agents, strong bases, and reactive metals. Store in a cool, dry, well-ventilated area. Do not store in aluminum, iron, or galvanized containers — lactic acid is corrosive to these metals.
Recommended storage temperature: 15–30 °C. Avoid freezing (may cause crystallization of concentrated solutions). Shelf life: 24 months in original sealed containers. Protect from direct sunlight and heat. Keep containers tightly sealed to prevent moisture loss and concentration change.
For more detailed information including pricing, customization, and shipping:
Discover more about this product and understand how it may fit into your operations, formulations, or production requirements.
Browse Product